Collagen & Quality
Clinical Study
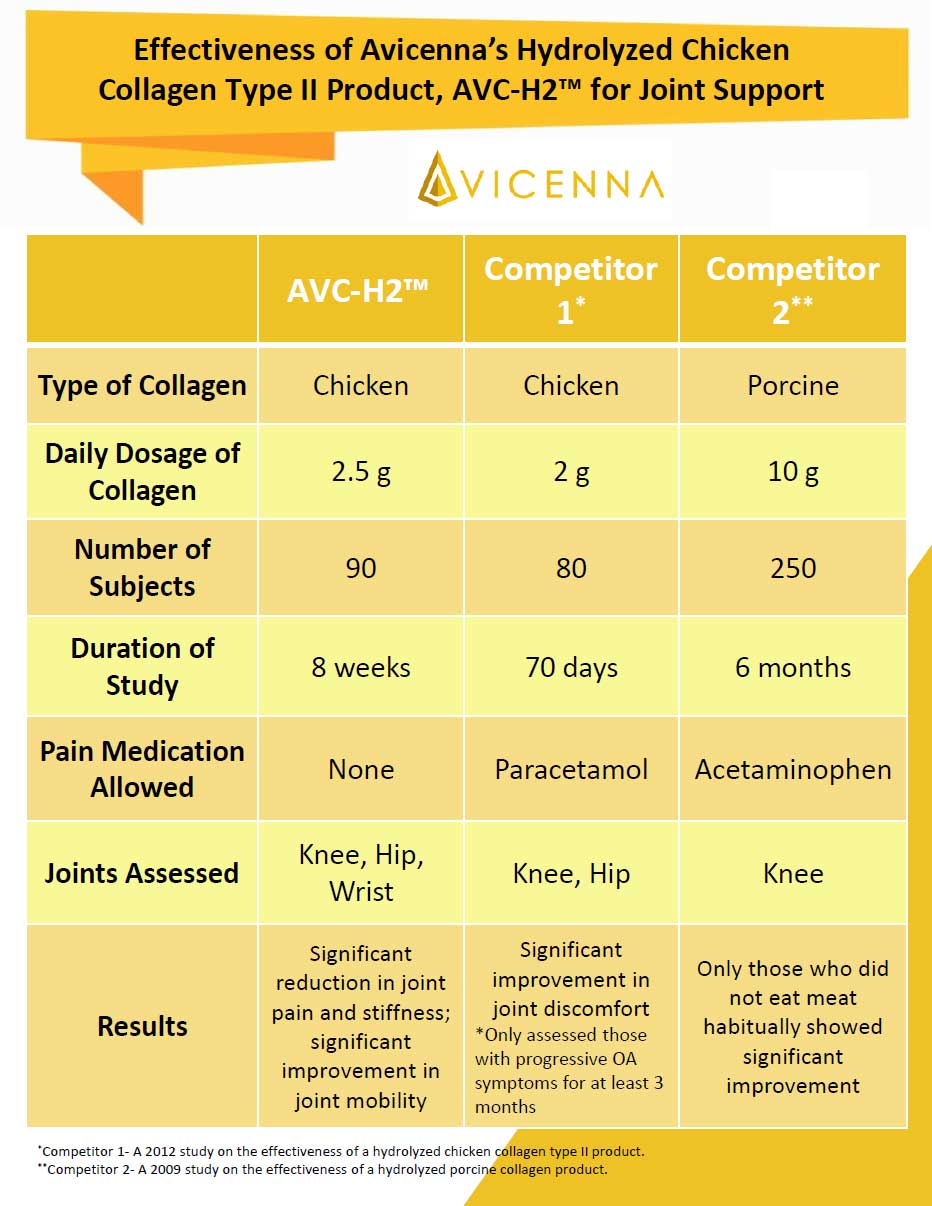
Avicenna Nutraceutical completed a double-blind, randomized, placebo-controlled clinical trial on its patented Hydrolyzed Chicken Collagen Type II raw material product, AVC-H2. The focus of the trial was to evaluate the efficacy of AVC-H2 in alleviating joint discomfort in the hip, knee, and wrist.
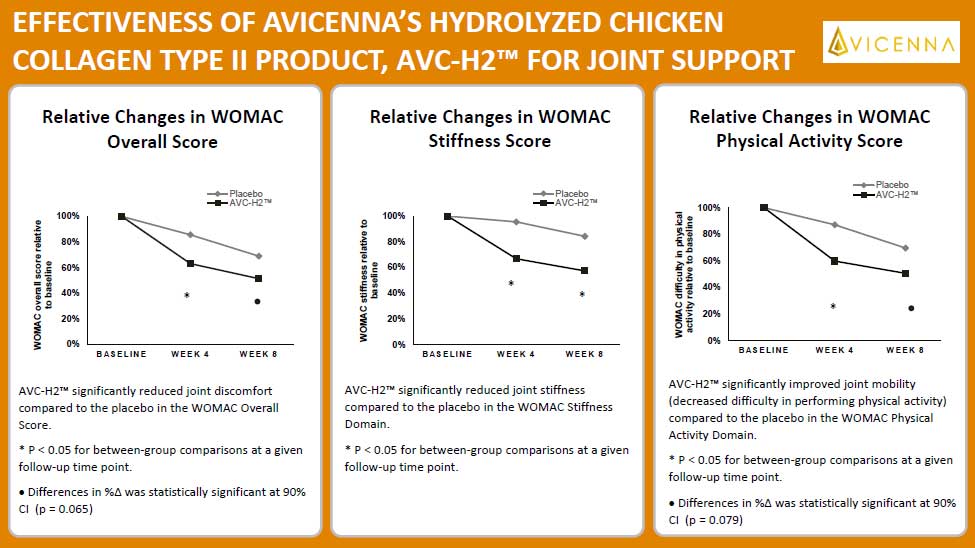
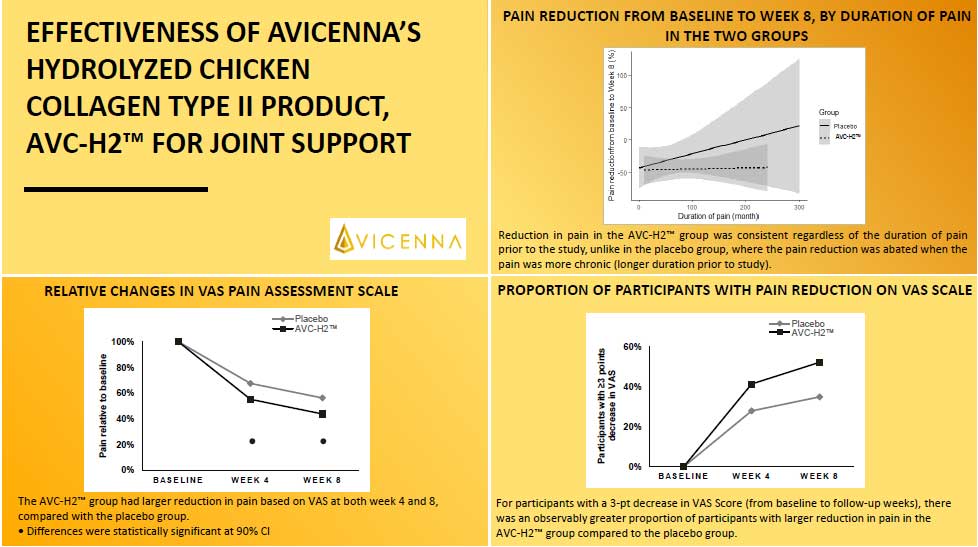
The trial was conducted for 8 weeks and included 90 individual participants. After 8 weeks, individuals who consumed AVC-H2 had a significant reduction in joint pain and stiffness and a significant increase in joint mobility in the hip, knee, and wrist compared to individuals who consumed the placebo.
Furthermore, Avicenna did not allow individuals to consume pain medication during the trial process because the use of pain medication would confound the results of the study. Other comparative studies of collagen products allowed their subjects to consume pain medication with the collagen test product. Those types of studies do not offer an accurate depiction of the results because it would be unclear whether test subjects benefited from the collagen or from the pain medication.
With AVC-H2, our customers can be assured that it is an effective joint support product based on the clear and significant results from the clinical trial.
The completed study was published in the Nutrients Journal!
Avicenna completed self-affirmed GRAS (Generally Recognized As Safe) notification for AVC-H2.